Plasma Donation by Shincheonji Church Facilitates Development of the Vaccine for COVID-19
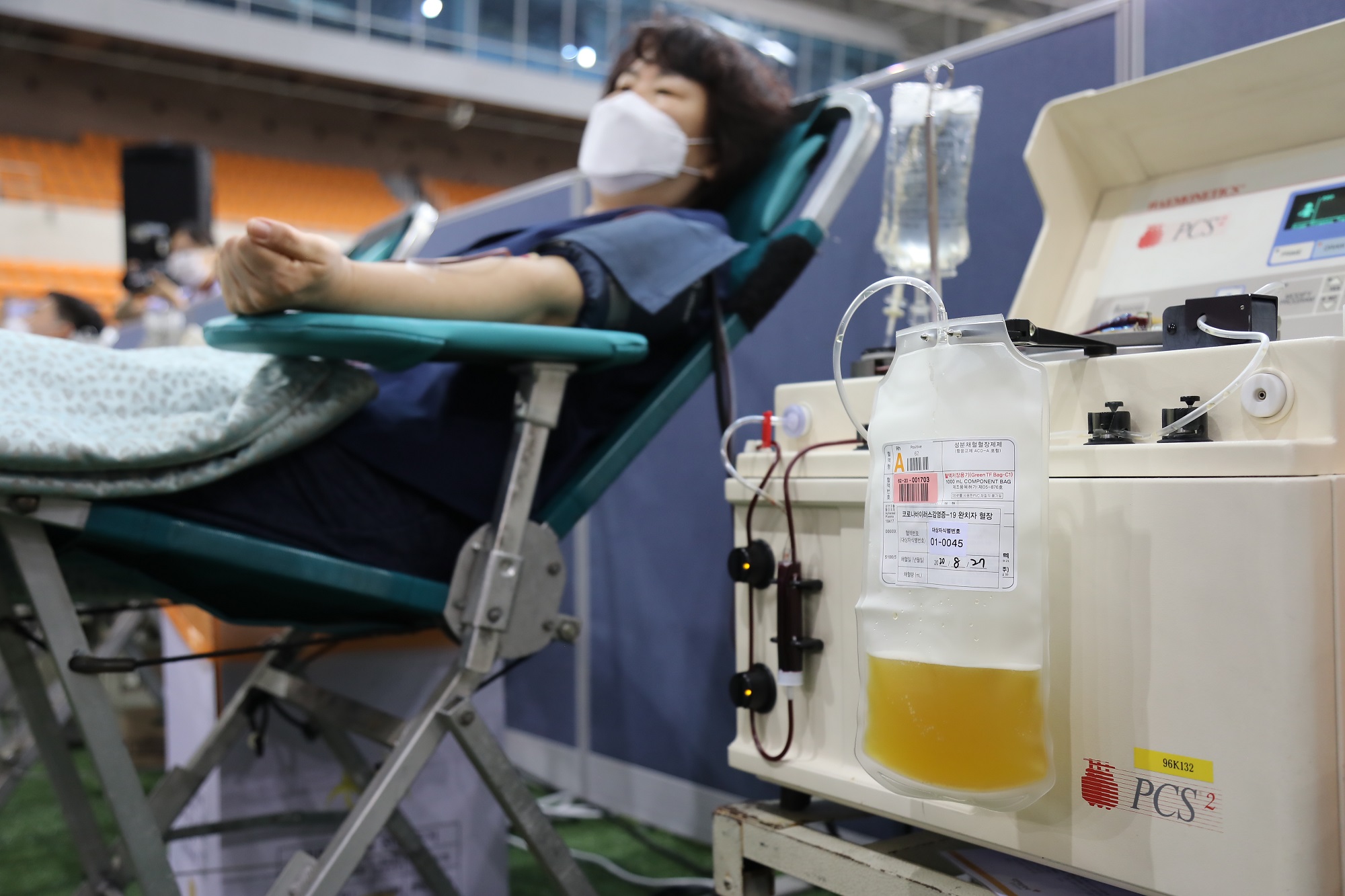
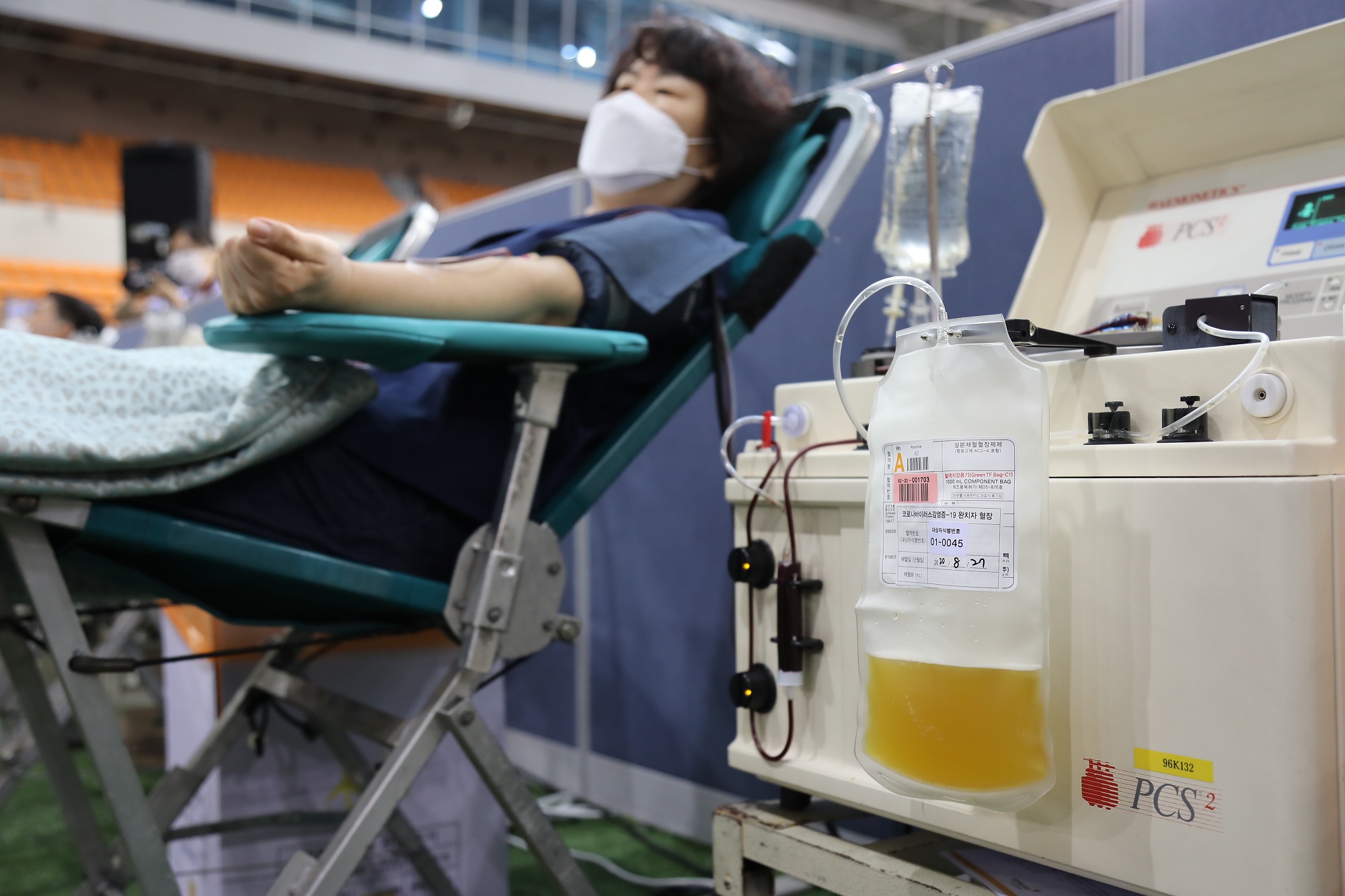
South Korea (GNP): On August 27th, over 1,000 members of religious organization called Shincheonji Church of Jesus participated in donating plasma for the cure of COVID-19.
This is carried out under the invitation of the health authorities in South Korea back on 24th, asking Shincheonji Church to cooperate for donating additional plasma for the development of the vaccine.
Early this year, around 5,000 confirmed cases were found in Shincheonji Church members with most infections from the city of Daegu, while most of them recovered from the virus with 11 deaths.
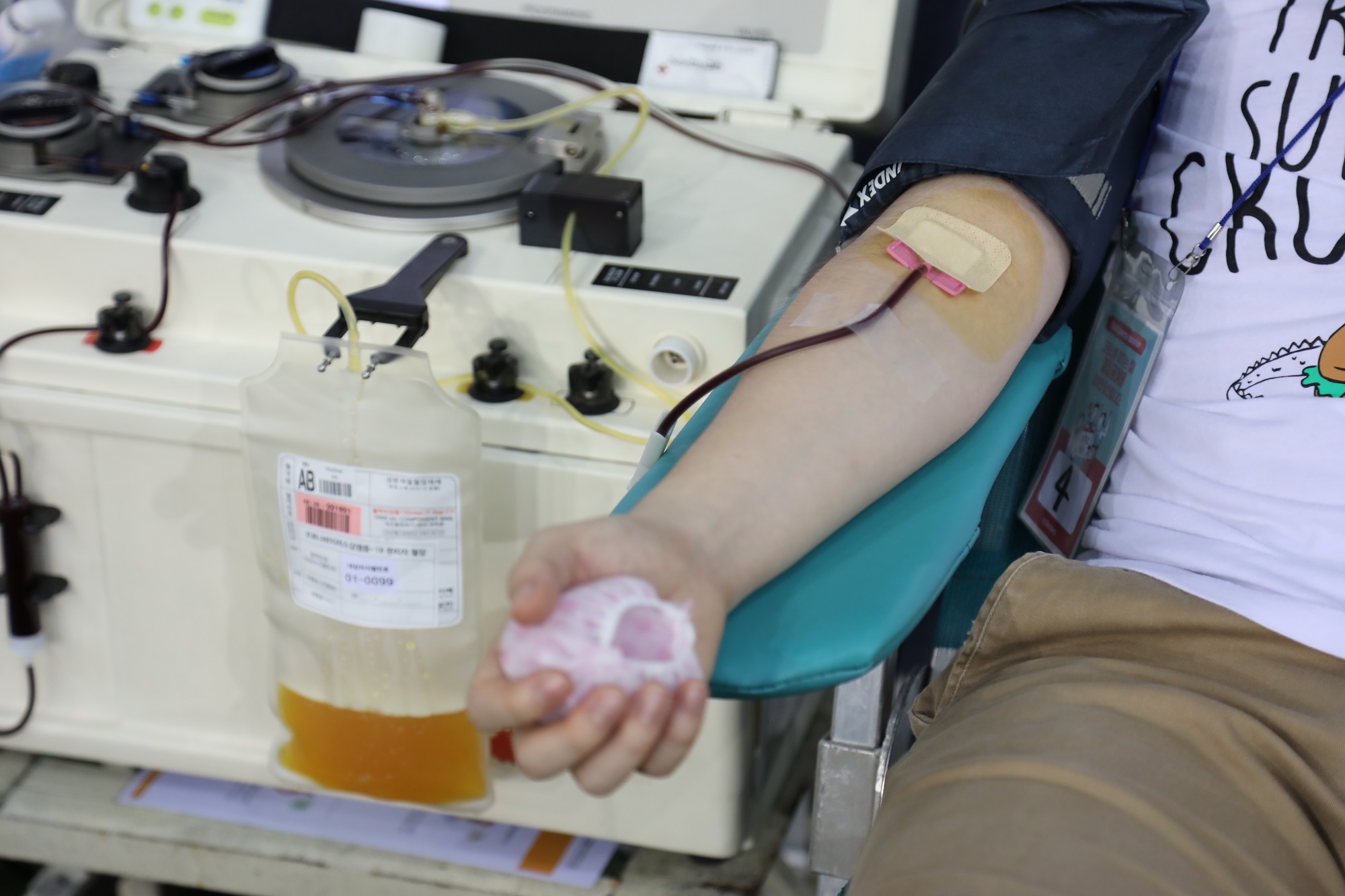
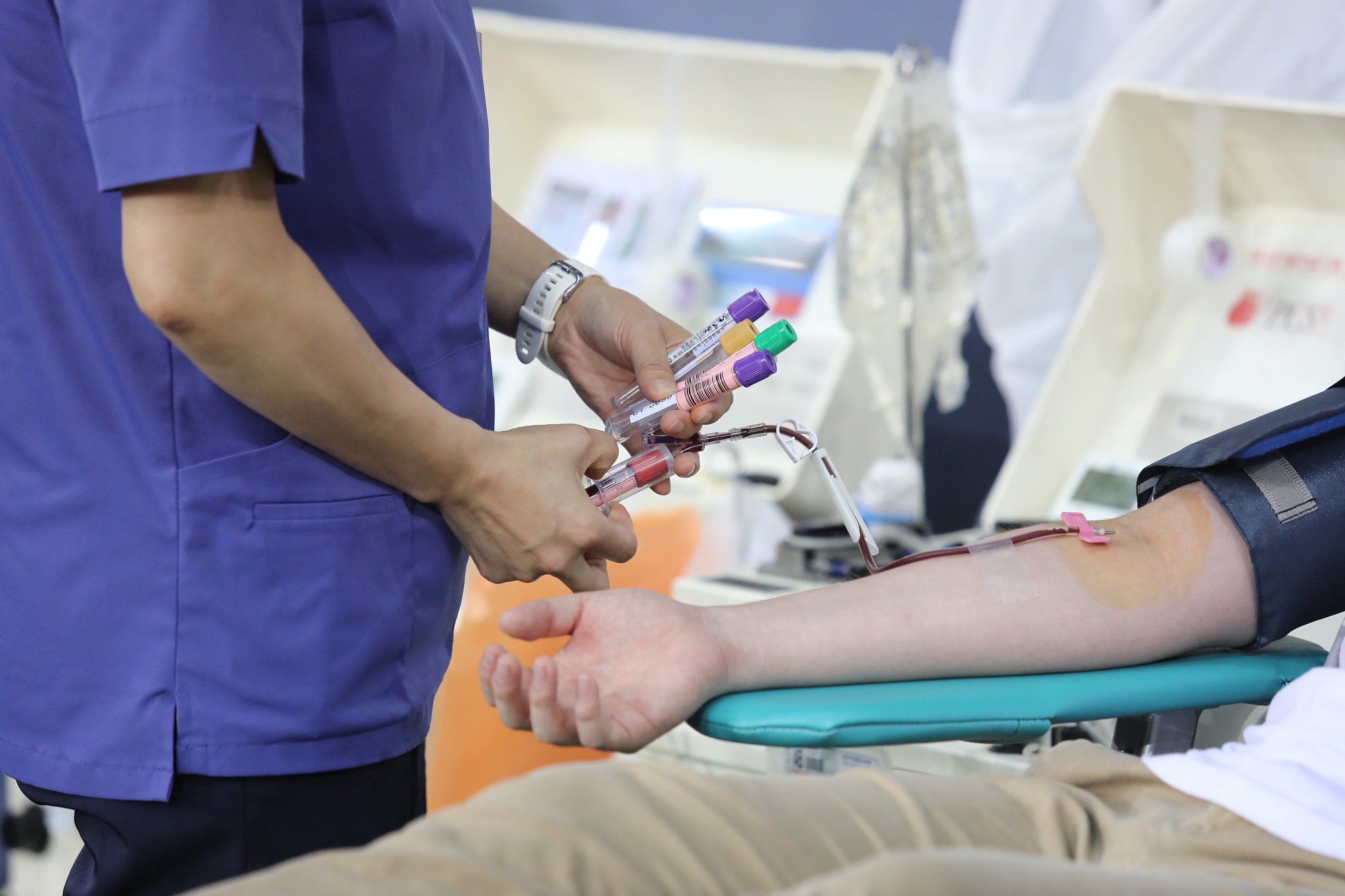
According to the report from the Korea Centers for Diseases Control and Prevention (KCDC), 562 recovered members from the church registered to donate plasma and 409 completed donation in July through the cooperation between KCDC and Shincheonji Church. Facing the need for “facilitation of developing a cure through plasma donation and clinical trial”, the KCDC asked another round of a large-scale donation by members of Shincheonji Church in August.
The KCDC in its official document expressed appreciation to Shincheonji Church (Chairman Lee, Man Hee) for “active participation in the collection of plasma as a group for the development of corona(virus) cure for the purpose of national health safety under the global crisis caused by the COVID-19.”
“With the cooperation of Daegu city, Daegu Athletics Center plans to provide the space, and GC Pharma plans to provide the necessary equipment and personnel from 27th August to 4th September. We express our gratitude to the city for providing a location for the group donations. We also express our gratitude to the congregation members of the religious organization, Shincheonji to be specific,” said Mr. Kwon Jun-wook, Deputy Director of the KCDC.
Research and development of the convalescent plasma treatment is underway by National Institute of Health under the Ministry of Health and Welfare in cooperation with Green Cross (GC) Pharma, a biotechnology company in South Korea.
On the same day, the US Food and Drug Administration (FDA) authorized an emergency use of convalescent plasma for the treatment of COVID-19. Experts say that more data need to be collected to see the benefit of such treatment.
Major challenges of researching the effectiveness and the development of convalescent plasma treatment come from limited supply from donors who must be recovered from the virus.
As a religious leader, Chairman Lee, Man Hee of Shincheonji in July said, “This (plasma donation) is the work that needs to be done as citizens of this country and as true believers. It is keeping the command of Jesus of loving your neighbor as yourself (Mt 22:39 of the Bible).”